The Hepatitis C Content Hub Channel
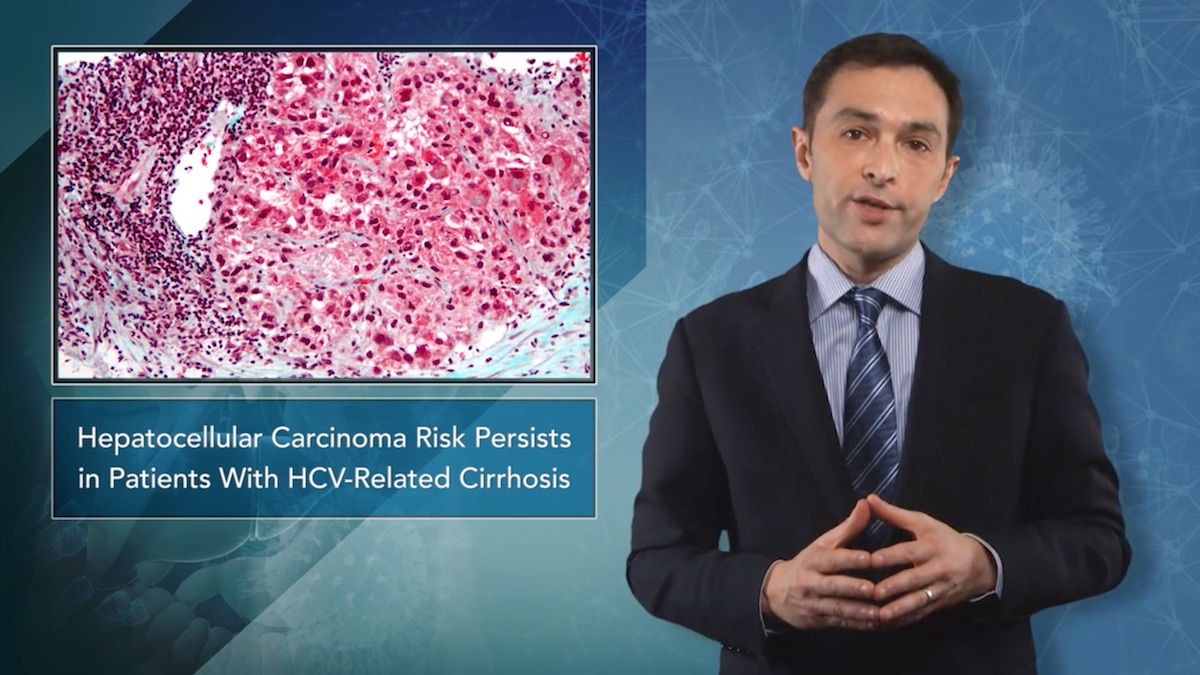
Infection-related deaths from hepatitis C virus (HCV) have a high prevention rate if patients have access to direct-acting antiviral (DAA) treatments. Relaxing the eligibility criteria by widening the program across the US may reduce the health and economic burden of HCV-related mortality.
References:
1. Kapadia SN, Jeng PJ, Schackman BR, Bao Y. State Medicaid hepatitis C treatment eligibility criteria and use of direct acting antivirals. Clin Infect Dis. 2017 Dec 1. doi: 10.1093/cid/cix1062. [Epub ahead of print] https://academic.oup.com/cid/advance-article/doi/10.1093/cid/cix1062/4682615
2. a) National Viral Hepatitis Roundtable and the Center for Health Law and Policy Innovation. Hepatitis C: the state of Medicaid access. National Summary Report. Published 14 November 2016. Washington, DC, 2016. https://www.chlpi.org/harvard-law-school-national-viral-hepatitis-roundtable-announce-preliminary-findings-in-project-to-grade-medicaid-access-to-hepatitis-c-treatment/
3. b) National Viral Hepatitis Roundtable and the Center for Health Law and Policy Innovation. Hepatitis C: the state of Medicaid access. National Summary Report. Published 23 October 2017. Washington, DC, 2017 http://nvhr.org/content/webinar-%E2%80%93-hepatitis-c-and-state-medicaid-access
4. Liao JM, Fischer MA. Restrictions of Hepatitis C Treatment for Substance-Using Medicaid Patients: Cost Versus Ethics. Am J Public Health. 2017;107:893-899. http://ajph.aphapublications.org/doi/abs/10.2105/AJPH.2017.303748?url_ver=Z39.88-2003&rfr_id=ori%3Arid%3Acrossref.org&rfr_dat=cr_pub%3Dpubmed&