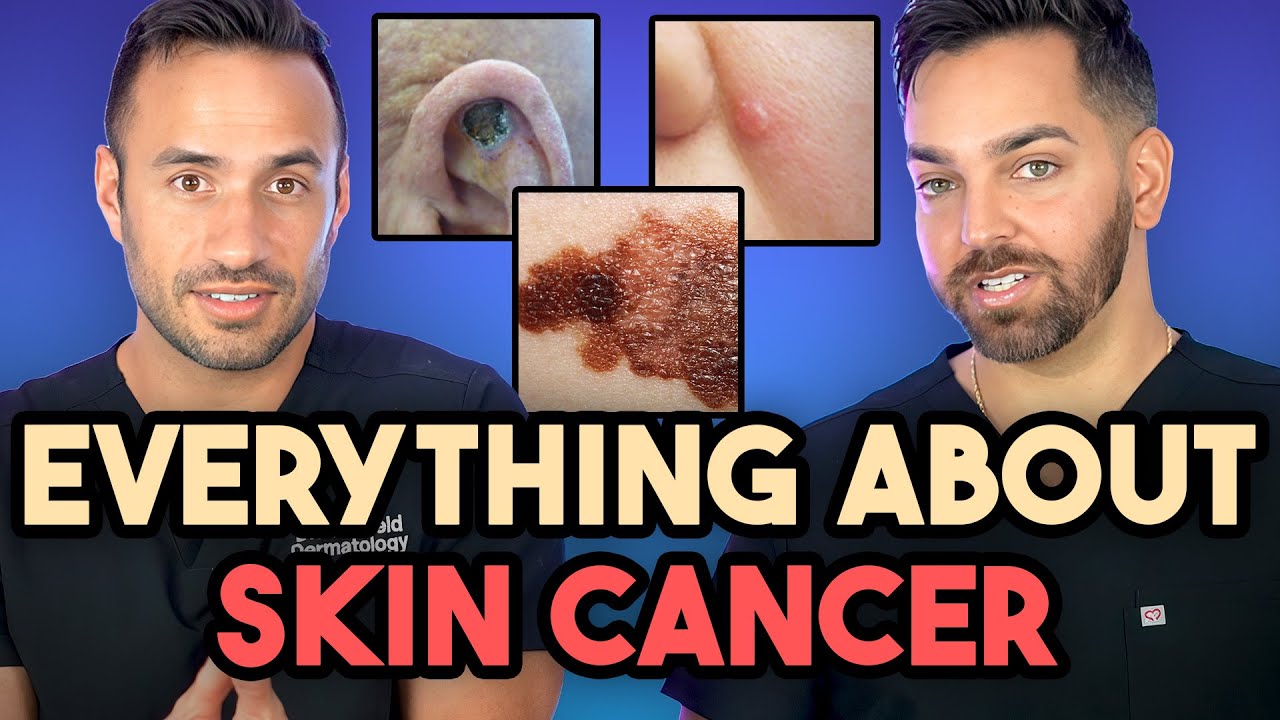
Drug combo not recommended for metastatic melanoma
Reuters Health • The Doctor's Channel Daily Newscast
“Despite a lack of prospective confirmation, the (thalidomide plus temozolomide) combination regimen became a community standard for patients with unresectable metastatic melanoma,” lead author Dr. Joseph I. Clark and co-researchers note. “Our intent was to expand on these results to better define the tolerability and efficacy of a set dose and schedule of the combination…in the cooperative group setting.”
In a Southwest Oncology Group trial, Dr. Clark, from Loyola University Medical Center, Maywood, Illinois, and associates assessed response rates and survival in 62 patients who were treated with thalidomide plus temozolomide. The eligibility criteria included stage IV disease, no active brain metastases, Zubrod performance score of 0 or 1, adequate organ function, and up to 1 prior systemic therapy excluding the study drugs and dacarbazine.
For patients younger than 70 years, thalidomide was given at a dose of 200 mg/d escalated to 400 mg/d, while for older patients, the drug was started at 100 mg/d and increased to 250 mg/d. Temozolomide was given at a dose of 75 mg/meters-squared per day for 6 weeks, followed by 2 weeks of rest, repeated until progression or toxicity.
The treatment response rate was 13%, and all of the responses were partial. This outcome, the authors note, is comparable to what would be expected with temozolomide or dacarbazine alone.
Progression-free survival at 6 months was 15%, and the estimated 1-year overall survival was 35%. The median overall survival was 8 months. No link was noted between progression-free or overall survival and a variety of biomarkers (e.g., interleukins, tumor necrosis factor).
One treatment-related death, due to MI, occurred, the report indicates. Other serious adverse events included one case each of pulmonary embolism, neutropenia, and central nervous system ischemia.
“On the basis of these results, use of this combination regimen is not recommended nor is further evaluation in a phase 3 trial appropriate,” the authors conclude.
Reference:
Cancer 2009.